
Magnesium can be found in chlorophyll molecules. You all know that calcium helps build your bones. The lighter alkaline earth metals, such as magnesium and calcium, are very important in animal and plant physiology. 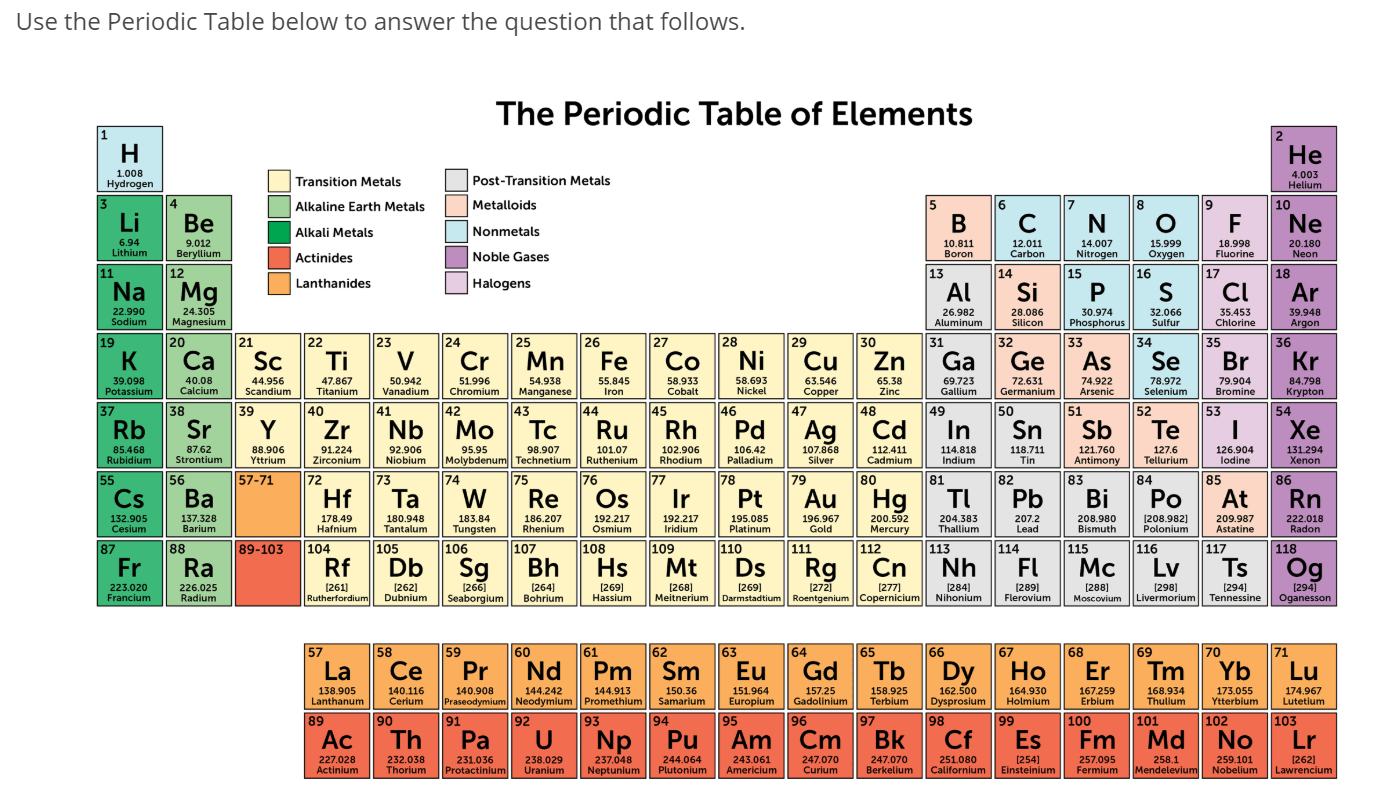 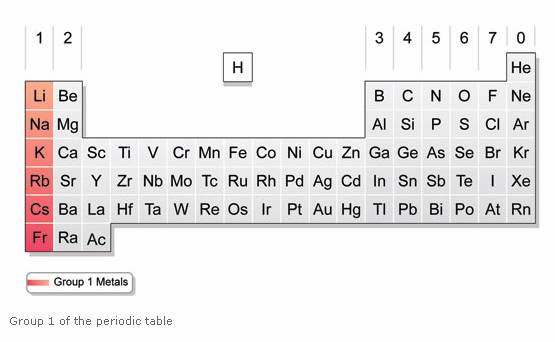
The other elements are found in many items, including fireworks, batteries, flashbulbs, and special alloys. It was originally mixed with zinc sulfide (ZnS). While radium is not found around your house anymore, it used to be an ingredient in glow-in-the-dark paints. It's all about giving up those electrons to have a full outer shell.Īs you get to the bottom of the list, you will find the radioactive radium. Sometimes you will see them with two halogen atoms, as with beryllium fluoride (BeF 2), and sometimes they might form a double bond, as with calcium oxide (CaO). They are ready to give up those two electrons in electrovalent/ionic bonds. Each of them has two electrons in their outer shell. While not as reactive as the alkali metals, this family knows how to make bonds very easily. As with all families, these elements share traits. Who's in the family? The members of the alkaline earth metals include: beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba) and radium (Ra). Those higher pH levels means that they are defined as "basic" or "alkaline" solutions. Do you know why they are called alkaline? When these compounds are mixed in solutions, they are likely to form solutions with a pH greater than 7. This is the second most reactive family of elements in the periodic table. You will find the alkaline earth metals right next door in Group II. The movement of electrical impulses through nerve fibers and the contraction of muscles are both mediated by the ions Ca 2+ and Mg 2+.So we just covered the alkali metals in Group I.They form alkaline solutions when they react with water. Beryllium is used in the manufacture of alloys. The group 1 metals are known as the alkali metals.By using barium, vacuum tubes can be made airless.Radium is employed for cancer treatment.Ca 2+ ions are essential components for the formation of bones and teeth.Magnesium, an important constituent of chlorophyll in green parts of plants takes part in photosynthesis.Reaction of alkaline earth metals with Oxygen Reaction of alkaline earth metals with water Reaction of alkaline earth metals with dilute HCl Reaction of alkaline earth metals with dilute H 2SO 4 Reaction of alkaline earth metals with Halogen Reaction of alkaline earth metals with liquid ammonia Uses of Alkaline earth metals On descending within the group, the elements in group 2 become more electropositive.Ĭalcium, strontium, and barium react similarly to sodium but are somewhat less reactive.Įxcept for beryllium, all metals create oxides in the air at ambient temperature. When added to water, both can form solutions having higher pH values (>pH). Alkaline earth metals are the elements in the group 2. The chemical behavior of alkaline earth metals is characterized by their great reducing power, which allows them to produce bivalent cations (M 2+) extremely quickly. Alkali metals are the elements in the group 1 of the periodic table. Physical Properties of Alkaline earth metals ElementsĬhemical Properties of Alkaline earth metal relatively low melting and boiling points.Ītomic Properties of Alkaline earth metals Elements.Some of the common properties of Group 2 elements are: Ultimately, each element in alkaline earth metals is in a +2-oxidation state. These elements have the general electrical configuration: ns 2Īlkaline earth metals have full s-orbital in their respective valence shells, so they quickly can lose two electrons to create +2 cations. Also, the name “ earth” refers to that group 2 metals, like calcium and magnesium, are among the prevalent elements of the planet’s crust. The term “ alkaline” refers to the fact that when these elements combine with oxygen, they produce basic compounds. The name kalium was taken from the word alkali, which came from Arabic al qali meaning the calcined ashes.The name potassium was made from the English word potash, meaning an alkali extracted in a pot from the ash of burnt wood or tree leaves. This group includes the elements beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra). What are Alkaline earth metals?Īlkaline earth metals are the elements that belong to group 2 of the periodic table i.e., the second vertical column. The physical and chemical properties of the elements in this group are very similar as the main objective of the periodic table is to study the elements and their compounds in a structured way and organized manner. These are the Group 2 elements in the periodic table. Alkaline earth metals are composed primarily of six elements: beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
